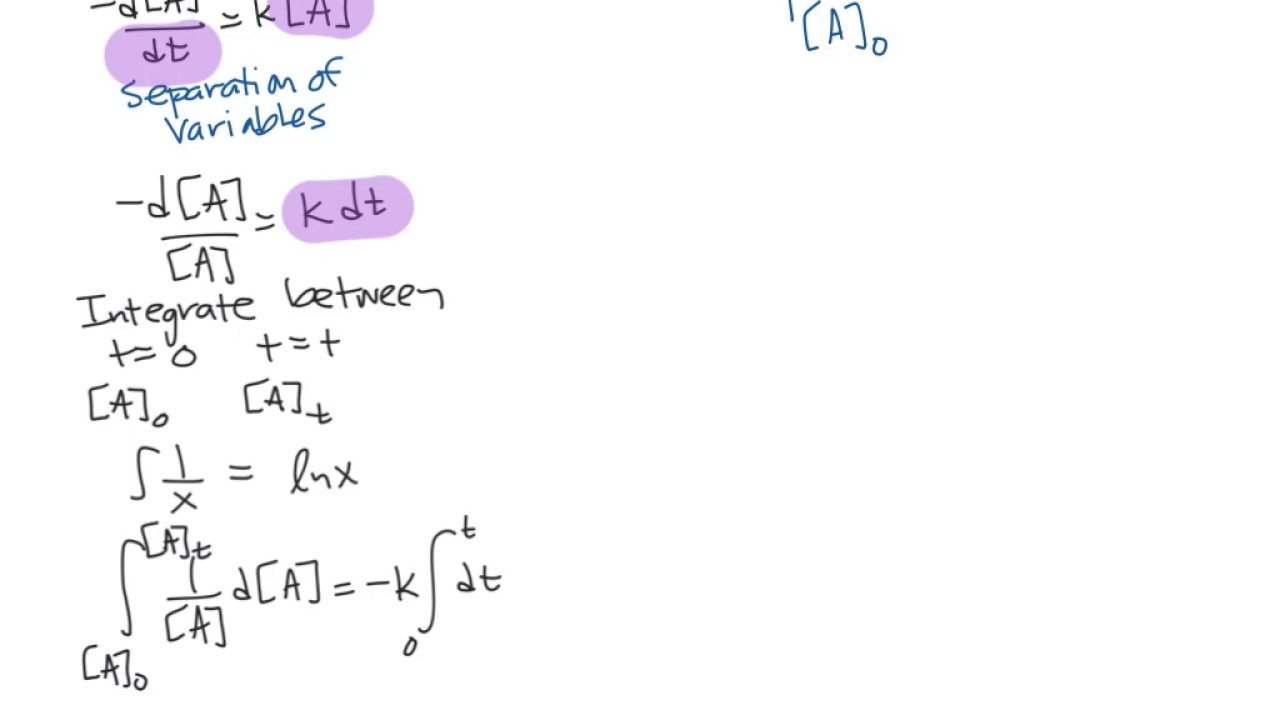For a first order reaction, the graph ln [a] vs time is a. The rate law calculator has rate of reaction functions for zero order, first order and second order reactions as follows: Zero order rate law (integral form) zero order half life zero order rate law first order rate law (integral form) first order half life first order rate law second order rate law (integral form) second order half life second order rate law the science.
Derive Integrated Rate Equation For First Order Reaction
[ a] t = − k t + [ a] 0 y = m x + b.
Integrated rate law equation for the chemical reaction a → b + c, integrated rate law can be expressed as a mathematical expression as given below.
The initial concentration of r be [r] 0, at t=0 Kt = 2.303log([r 0]/[r]) (or) k =. We can use integrated rate laws with experimental data that consist of time and concentration information to determine the order and rate constant of a reaction. (something weird happening with the camera.
Where i is the constant of integration.
The integrated rate law can be rearranged to a standard linear equation format: The integrated rate law can be rearranged to a standard linear equation format: Where, [r 0] is the initial concentration of the reactant (when t = 0) [r] is the concentration of the reactant at time ‘t’ k is the rate constant; An integrated rate law comes from an ordinary rate law.
The first order rate integral[1] equation calculates the rate at which the reactants turn in to products.
We know that the rate law is the expression in which reaction rate is given in terms of molar concentration of reactants with each term raised to some power, which may or may not be equal to the stoichiometric coefficient of the reacting species in a balanced chemical equation. 2a products or a + b products (when [a] = [b]) , rate = k[a] 2 the integrated rate law is 1/[a] = kt + 1/[a o ] The value of i can be calculated from the initial concentration. We get the following when we use the resultant value of c in equation (1):
[ a] t = − k t + [ a] 0 y = m x + b.
See what is the rate law?. I'll get a new one.)how does concentration of reactant change with time when rate=k[a]^3? These rate laws help us determine the overall mechanism of reaction (or process) by which the reactants turn into products. The above equation is known as integrated rate equation for zero order reactions.
A full integration of the equation can be found here.
Namely, the concentration versus time data are fit to the following equation: As this can be easily graphed. And by distinguishing the patterns of the graphs, we can quickly determine if the reaction is 0th, 1st, or 2nd order. Rate laws from graphs of concentration versus time (integrated rate laws) in order to determine the rate law for a reaction from a set of data consisting of concentration (or the values of some function of concentration) versus time, make three graphs.
The integrated rate equation for the main product [c] is [] = [] [′ [] (′)], which is equivalent to [] [] [] = [] ′ (′).
Y = mx + c. These are inherently differential equations, because the rate is always defined as a change in concentration with time; Thus, the graph of the second order integrated rate law is a straight. The equation for the second order integrated rate law takes the form y = mx +b, where y = 1/a;
𝑅 p =− [ ] = g[ ] on the other hand, integrated rate laws express the reaction rate as a function of the initial concentration and a measured (actual) concentration of one or more reactants after a specific amount of time (t) has passed;